- Home
- Medicinal Chemistry
- small molecule drug discovery
High-Throughput Screening (HTS)
HTS is a method used in drug discovery and related fields of research. Using robotics and the appropriate software, high-throughput screening allows a researcher to quickly conduct large numbers of in vitro assays against large libraries of distinct chemical entities to identify active compounds that modulate a molecular pathway. The collection of total HTS "hits"represents a starting point for HTS triage. HTS triage involves the classification, or prioritization, of hits from screening campaigns into compounds that are likely to survive further investigation, those that probably have no chance of succeeding as probes (tool compound) and those that fall into an intermediate classification where intervention could make a significant difference in their survival. Compounds or series of compounds are prioritized based on biochemical (promiscuity, structural alerts, etc.) and physciochemical (MW, cLogP, solubility, structural complexity, rule-of-5-compliance. etc) properties. The outcome of HTS is the identification of 1-3 compounds / series for further manipulation via synthesis or catalog purchases and would represent the initiation of lead development or could be a starting point for drug design and for understanding the interaction or role of a particular biochemical process in biology.
Services offered:
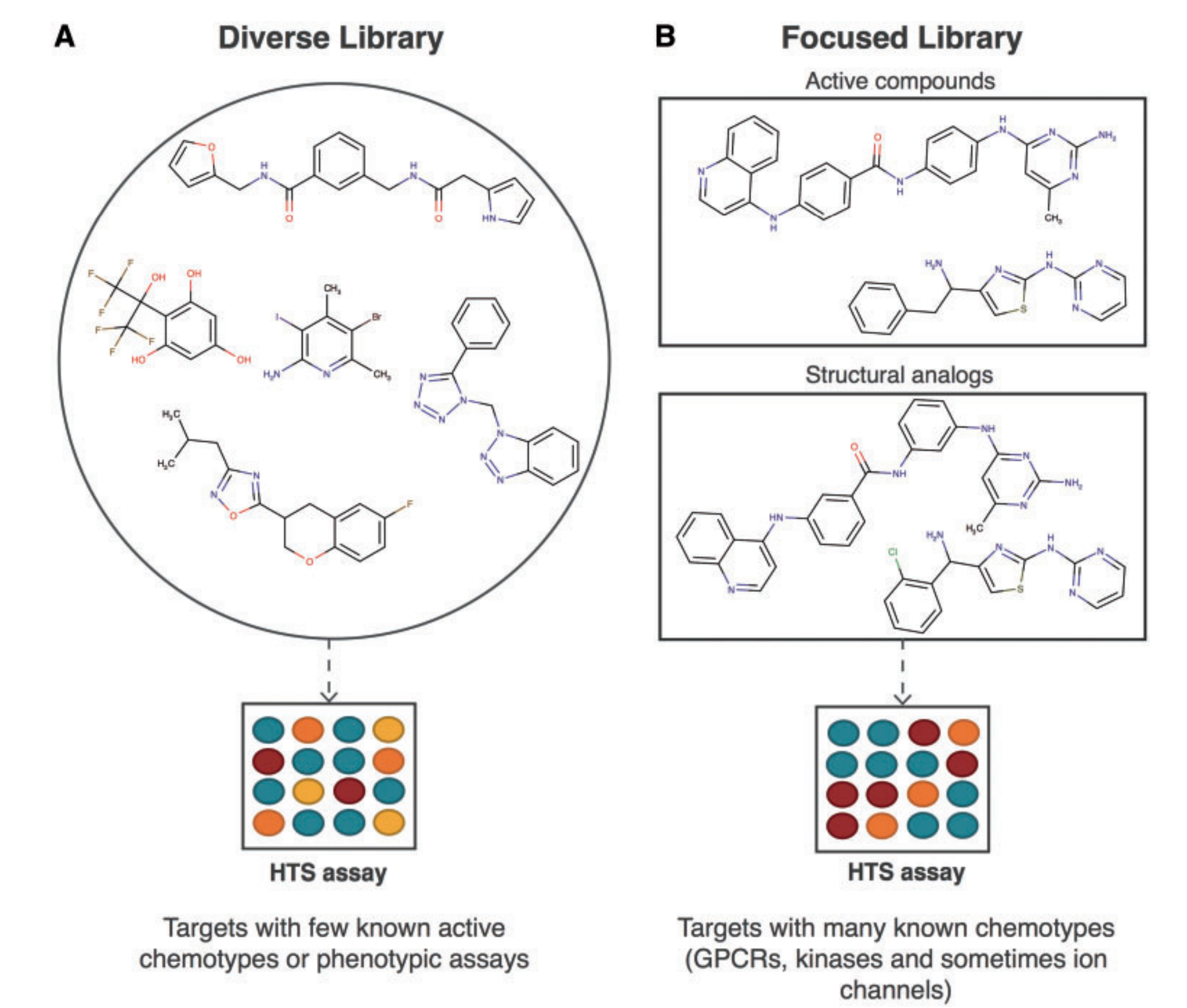
Diverse libraries compared with focused libraries diagram
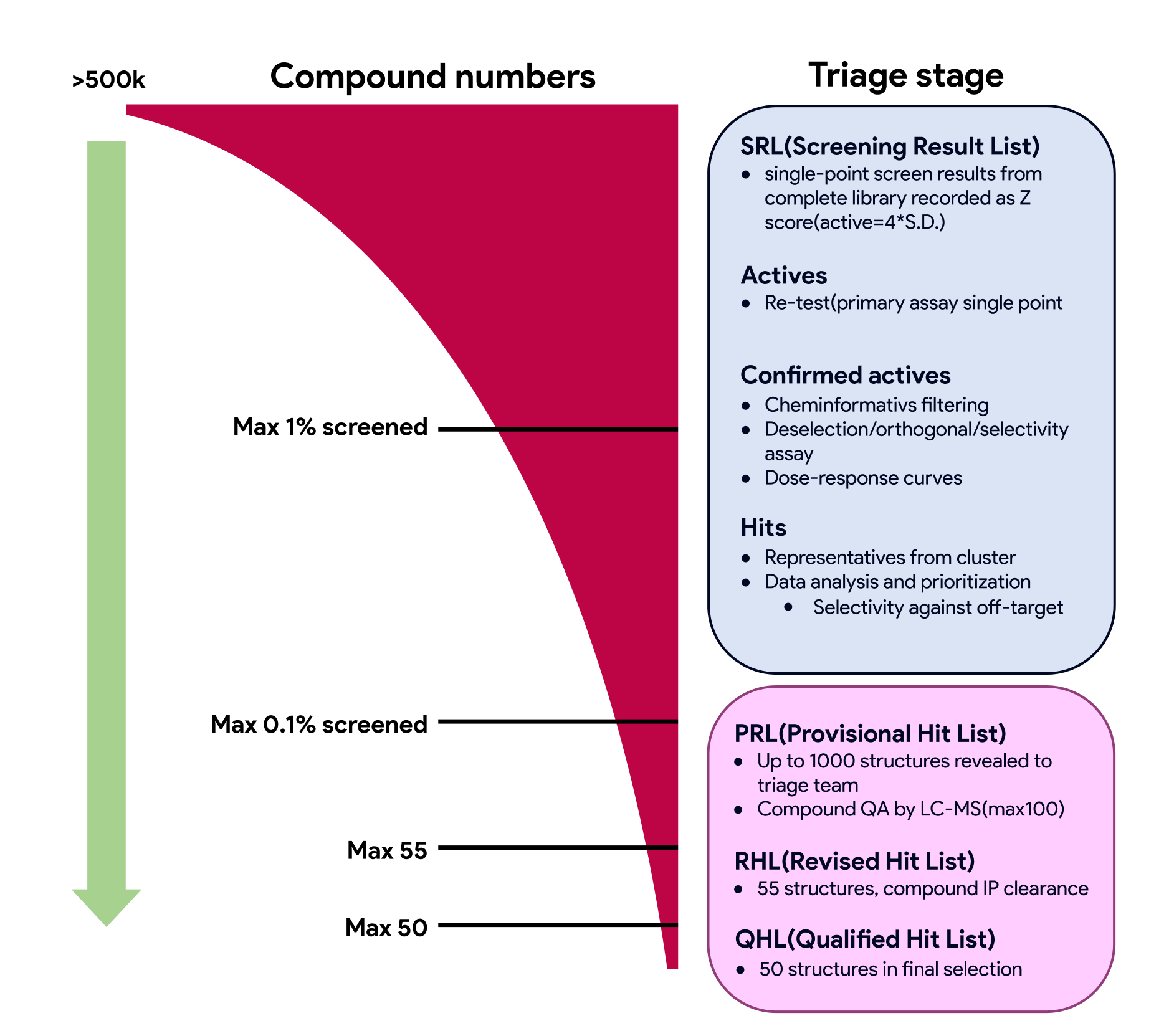
Representative visualization of the HTS triage process to generate the Qualified Hit List
Structure Based Drug Design (SBDD)
SBDD is a specific, efficient, and rapid process for lead discovery and optimization when the three-dimensional structure of the biological target is obtained through methods such as x-ray crystallography or NMR spectroscopy because it deals with the 3D structure of a target protein and knowledge about the disease at the molecular level. Among the relevant computational techniques, structure-based virtual screening (SBVS), molecular docking, and molecular dynamics (MD) simulations are the most common methods used in SBDD. These methods have numerous applications in the analysis of binding energetics, ligand-protein interactions, and evaluation of the conformational changes occurring during the docking process. This information is integrated to predict high binding affinity compounds which are subsequently prepared synthetically.
Services offered:
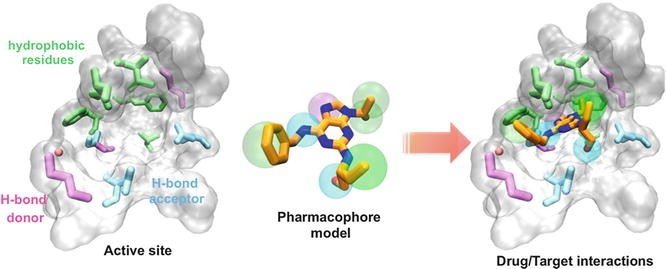
Structure Activity Relationship (SAR)
A structure—activity relationship (SAR) is the relationship between the chemical or 3D structure of a molecule and its biological activity. The analysis of SAR enables the determination of the chemical group or substitution responsible for a biological effect. The developed understanding allows further modulation of the effect of a bioactive compound by changing its chemical structure. Chemical synthesis is used to insert new chemical groups and test the modifications for their biological effects.
Services offered:

Scaffold-Hopping
Scaffold-hopping in drug-design is widely used in drug discovery. Scaffold-hopping requires an active molecule with either an experimentally determined, or a hypothetical, binding conformation. A central part or a portion of the molecule is then replaced by a new scaffold which is able to retain the original binding groups of the molecule in their optimized binding orientation. The process allows for the identification of new scaffolds which might impart better physicochemical properties or avoid existing patent art.
Services offered:
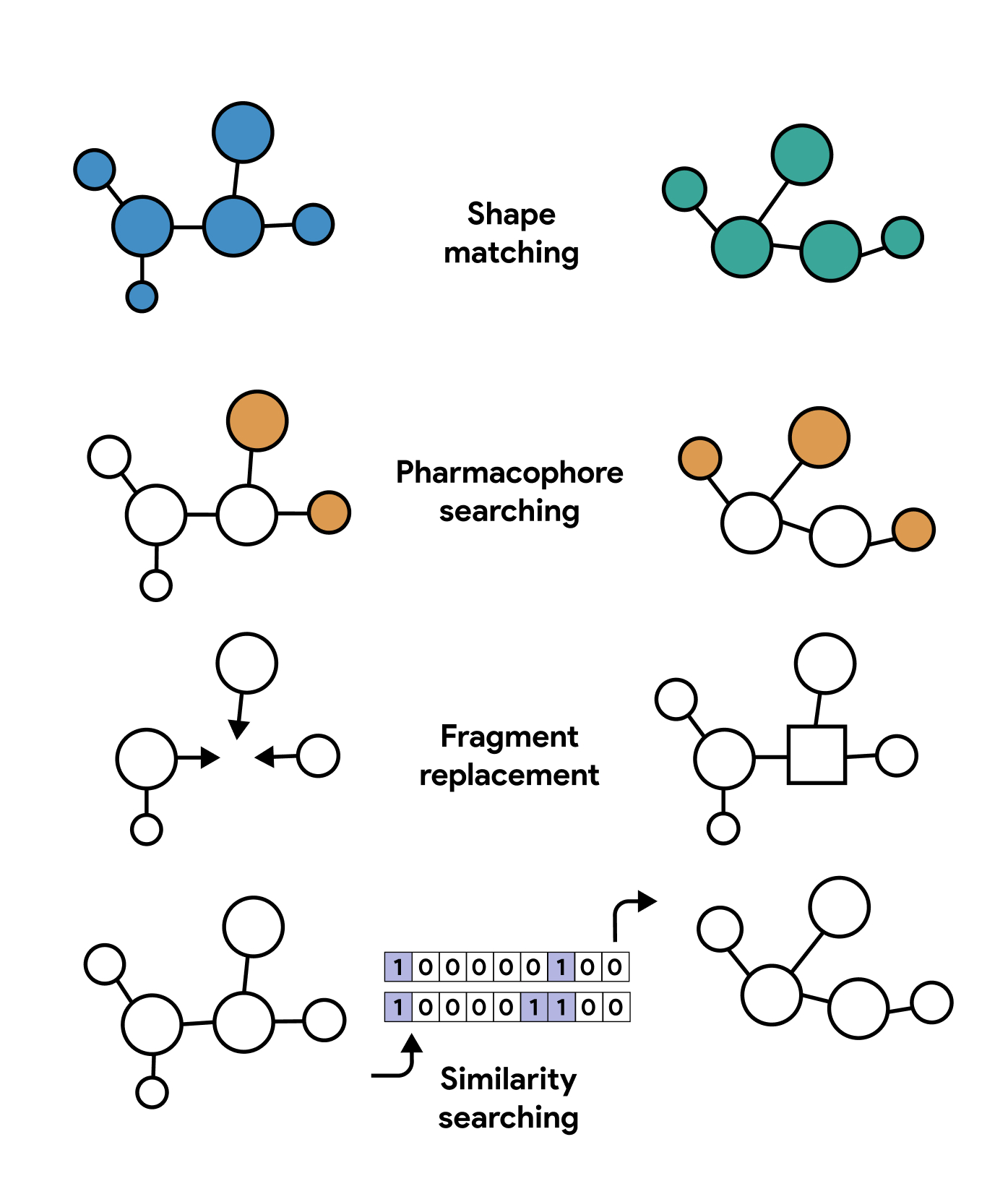
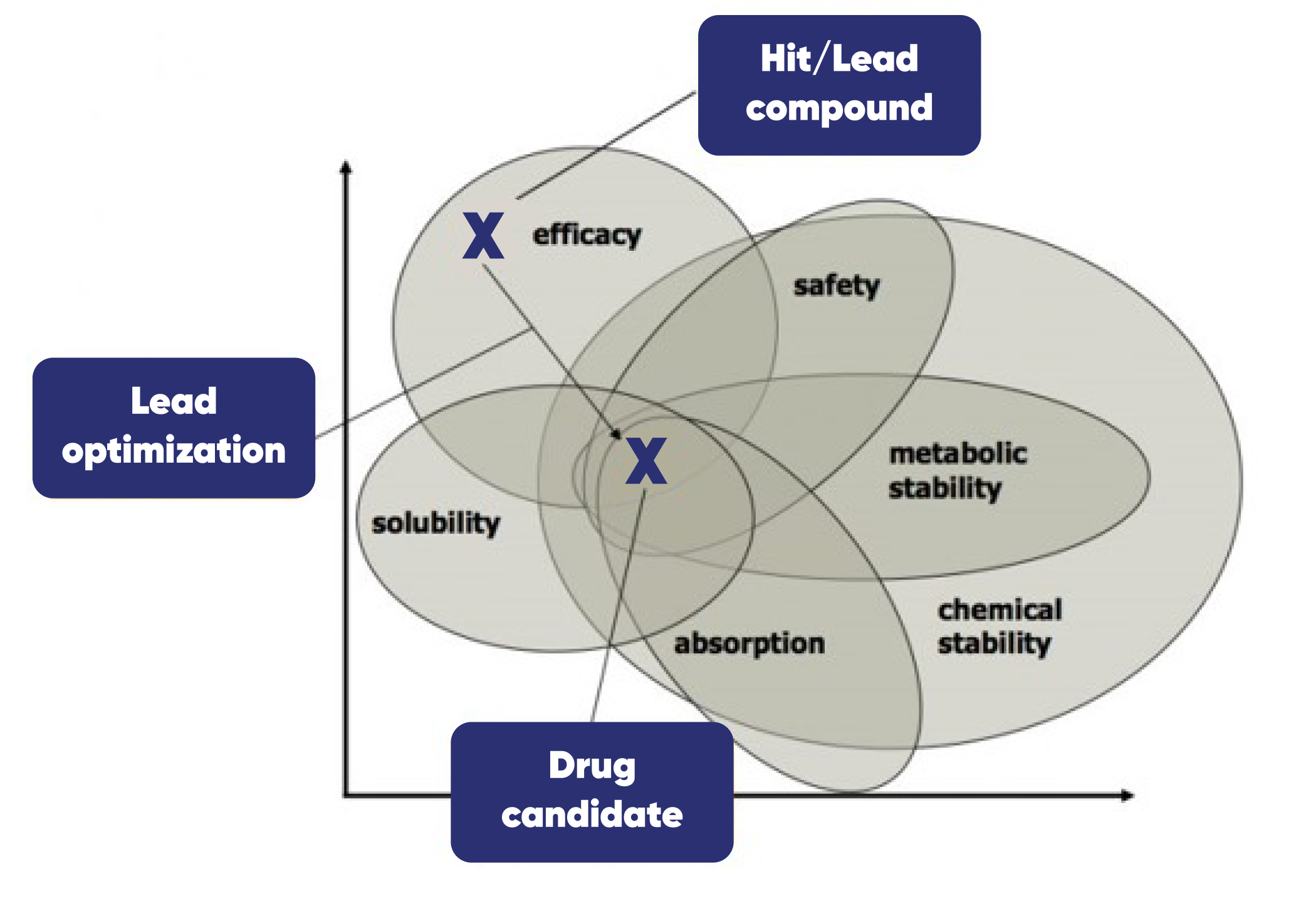
Lead Development
Lead development requires identification of a lead small molecule from HTS screening or similar with potential to be made into a clinical candidate. Once identified, the chemical structure of the lead compound is chemically modified to improve potency, selectivity or pharmacokinetic (PK) parameters.
Services offered:
Preparing compounds fast and efficiently requires deep expertise in a wide range of chemical transformations.
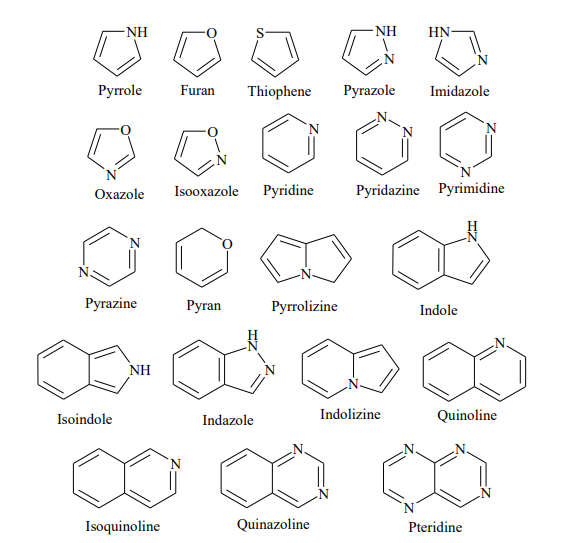
- Synthesis and decoration of C-, N-, O- and S-containing heterocycles
- Synthesis and derivatizations of fused-heterocyclic systems
- Saturated heterocyclic systems with C-N, C-O and C-P bonds
